
search
May 12, 2026
The evolution of targeted therapies has positioned antibody-drug conjugates (ADCs) among the most promising modalities in modern oncology. By combining the targeting precision of biologics with the cell-killing power of cytotoxic drugs, ADCs offer a more focused approach to cancer treatment.
As ADC development continues to accelerate, advances in both biology and chemistry are enabling safer, more effective therapies. These innovations are driving increased adoption of ADCs across the broader drug development landscape.
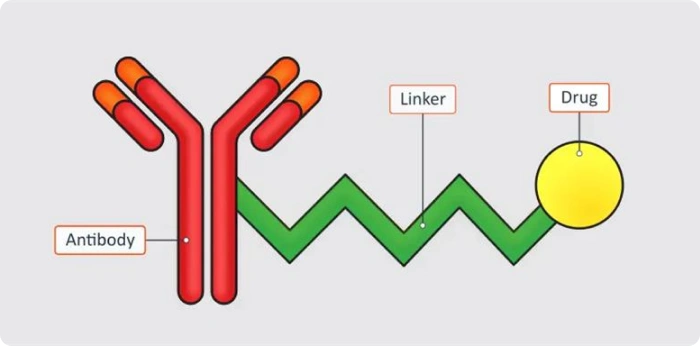

An ADC is a complex therapeutic composed of three core components: a monoclonal antibody, a cytotoxic payload, and a chemical linker that connects them.
The antibody is designed to recognize and bind to specific antigens expressed on cancer cells. This targeting capability allows ADCs to selectively deliver potent drugs to diseased cells while minimizing exposure to healthy tissue.
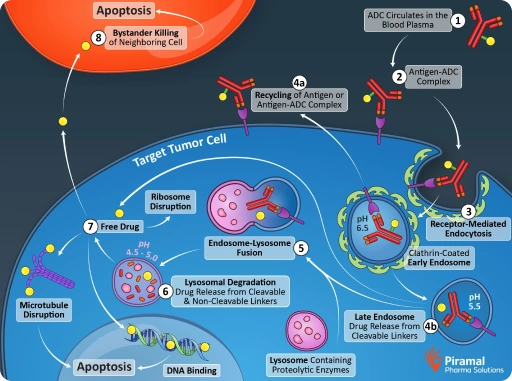
The mechanism of action for ADCs is highly coordinated. After binding to a target antigen, the ADC is internalized into the cancer cell. Once inside, the linker is cleaved (or the antibody is degraded), releasing the cytotoxic payload.
This targeted delivery enables the payload to disrupt critical cellular processes, ultimately leading to cell death. In some cases, released payloads can also affect neighboring tumor cells, enhancing therapeutic impact in heterogeneous tumors.

The effectiveness of ADCs depends on the careful integration of biology and chemistry. The biological component, the antibody, determines target specificity, binding affinity, and internalization efficiency.
On the chemistry side, payload-linkers play a critical role in ensuring stability and controlled drug release. Linkers must remain stable in circulation to prevent premature payload release while enabling efficient activation once inside the target cell.
Cleavable linkers respond to specific biological triggers such as pH or enzymatic activity, while non-cleavable linkers rely on intracellular degradation processes. Each approach offers advantages depending on the therapeutic context and tumor biology.


The chemistry of ADCs is a key driver of performance and manufacturability. Design considerations for payload-linkers include hydrophobicity, drug-to-antibody ratio (DAR), and conjugation strategy.
Small changes in linker structure can significantly impact pharmacokinetics, aggregation, and overall stability. The choice of payload, which is often a highly potent cytotoxic compound, must also balance efficacy with safety.
As ADC pipelines become more competitive, many developers are moving toward customized payload-linkers to differentiate their therapies and optimize clinical outcomes.
Developing ADCs presents unique challenges compared to traditional therapies. The integration of biologics production, chemical synthesis, and bioconjugation requires highly coordinated processes.
Manufacturing must also account for handling highly potent payloads, which require specialized containment and analytical capabilities. Ensuring consistency across batches and maintaining product stability are critical for regulatory approval and commercialization.
ADCs represent a powerful convergence of biology and chemistry in modern drug development. By enabling targeted delivery of highly potent therapies, they are transforming the oncology treatment landscape.
As innovation in payload-linkers and conjugation technologies continues, ADC development will play an increasingly important role in advancing precision medicine.
An antibody-drug conjugate (ADC) is a targeted therapy that combines an antibody with a cytotoxic drug to selectively kill cancer cells.
Payload-linkers are important in ADCs because they control drug stability and release, thereby impacting both safety and efficacy.
ADC development is complex because it requires integrating biologics, chemistry, and high potency manufacturing processes.
The following Piramal facilities support ADC development and manufacturing:
