
search
Apr 02, 2026
Antibody-drug conjugates (ADCs) have rapidly become a cornerstone of modern oncology, driven by significant advances in payload-linker technologies. While early ADC programs struggled with stability and manufacturing complexity, recent innovation has drastically improved their clinical viability. Today, payload-linker engineering plays a central role in determining not only therapeutic performance, but also scalability and manufacturability.
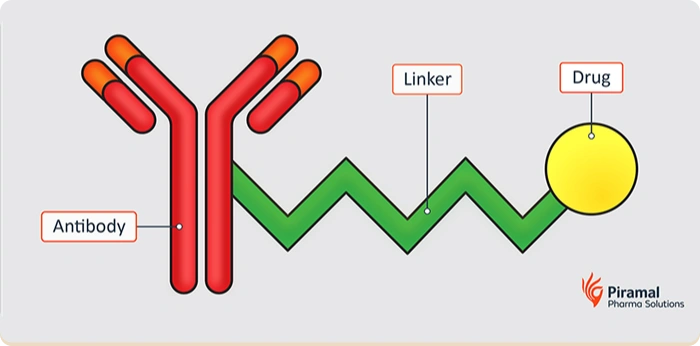

Although the core concept of ADCs — delivering a potent cytotoxic payload via a targeted antibody — remains unchanged, execution has advanced considerably. In recent years, the number of ADCs in clinical development has grown substantially, reflecting increased confidence in the modality and improvements in enabling technologies.
As pipelines expand, differentiation is no longer driven solely by the antibody. Instead, the design of the payload-linker system has become a key lever for optimizing therapeutic index, safety, and overall drug performance.
The linker is a critical component of an ADC, directly influencing pharmacokinetics, efficacy, and safety. It must remain stable in circulation while enabling controlled release of the payload at the target site.
Linkers are typically categorized as:
Beyond this classification, linker properties such as hydrophobicity, drug-to-antibody ratio (DAR), and conjugation compatibility significantly impact aggregation, clearance, and batch consistency. Even small design changes can have meaningful downstream effects.
As ADC development becomes more competitive, many sponsors are moving away from standardized constructs toward custom payload-linker designs. While this enables greater optimization, it also introduces manufacturing complexity.
Most payloads used in ADCs are highly potent active pharmaceutical ingredients (HPAPIs), requiring strict containment and specialized handling. Manufacturing these compounds safely demands:
Facilities must also support both early-phase development and later-stage scale-up, ensuring alignment with GMP expectations and regulatory requirements.
When fragmented development occurs, and payloads, linkers, antibodies, and drug product manufacturing are handled by separate vendors, inefficiencies and risks arise. As a result, many companies are shifting toward integrated development models.
An integrated approach enables parallel development across components, reducing timelines and improving coordination. It also supports smoother scale-up, better control of critical quality attributes, and reduced complexity during technology transfer.
This becomes especially important as ADCs grow more complex, incorporating new payload classes, targeting strategies, and conjugation chemistries.

Piramal Pharma Solutions supports ADC programs through our Riverview, Michigan site, which specializes in HPAPI and payload-linker development and manufacturing. The facility is equipped to handle both standard and highly potent compounds, with dedicated containment systems and purification capabilities.
With expertise in process development, scale-up, and regulatory support, we enable the production of custom payload-linkers tailored to specific program needs. Ongoing investments in high potency infrastructure and analytical technologies further strengthen our ability to support ADC programs from early development through commercialization.
Payload-linker engineering has become a defining factor in ADC development, influencing everything from clinical efficacy to manufacturing feasibility. As the field continues to evolve, success will depend on the ability to design differentiated constructs while ensuring scalable, compliant production. Integrated expertise across chemistry, manufacturing, and analytics will be essential to delivering the next generation of targeted therapies.
A payload-linker connects the cytotoxic drug to the antibody and controls how and when the drug is released at the target site.
Linker design affects stability, safety, and efficacy by determining how the payload behaves in circulation and within target cells.
Challenges associated with payload-linker manufacturing include handling highly potent compounds, ensuring containment, achieving impurity control, and scaling processes for GMP production.
The following facility provides comprehensive payload-linker development and manufacturing solutions.
