
search
Mar 24, 2026
Oxidation reactions play a central role in chemical synthesis, enabling medicinal chemists to functionalize molecules while controlling reactivity and selectivity. At manufacturing scale, however, oxidation chemistry presents safety and operational challenges that often push teams toward alternative transformations. Despite these constraints, some syntheses require oxidation reactions that cannot be easily replaced. When it can be executed safely at scale, ozonolysis stands out for its atom efficiency, clean reaction profile, and synthetic precision.
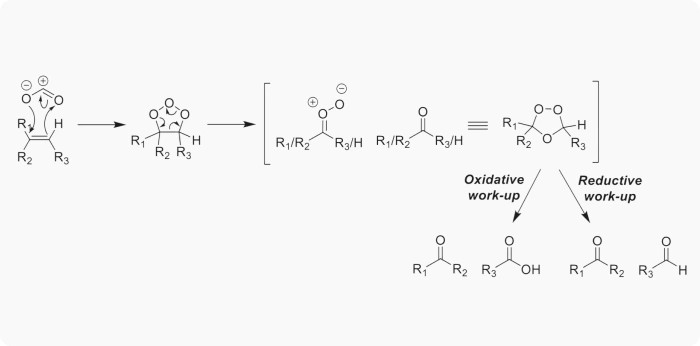

Ozonolysis is a well-established oxidation reaction that cleaves carbon–carbon double or triple bonds using ozone. The reaction produces aldehydes, ketones, carboxylic acids, or alcohols, depending on reaction conditions and quenching strategy. Its mechanism proceeds through a Criegee intermediate, which can be selectively quenched to achieve the desired functional group transformation.
Because of its predictable chemistry and versatility, ozonolysis is widely used at laboratory scale. Translating this reaction to API manufacturing requires additional safeguards, but the underlying chemistry remains highly attractive.
Compared with metal-mediated oxidations or peroxide-based systems, ozonolysis offers several advantages. Ozone enables precise cleavage with minimal side reactions, thereby reducing downstream purification burdens. The reaction avoids the use of stoichiometric amounts of heavy metals, eliminating metal contamination risks and reducing waste generation.
The gaseous nature of ozone also reduces solvent demand, supporting greener processing and improved economic efficiency. In addition, ozonolysis can often be performed under relatively mild thermal conditions, which helps protect sensitive intermediates from degradation – an especially important consideration in pharmaceutical manufacturing.


Despite its benefits, ozonolysis presents inherent scale-up challenges. The reaction generates unstable ozonides and peroxide intermediates that can be energetic or explosive if not properly controlled. Ozone itself is a highly reactive and toxic gas, and ozonolysis reactions are frequently exothermic.
Successful scale-up depends on careful control of temperature, gas–liquid mass transfer, reaction endpoints, and quenching strategies. Process calorimetry, peroxide monitoring, and robust safety assessments are essential. While ozonolysis has been implemented at multi-kilogram scale in the API industry, many facilities lack the specialized equipment and experience required to do so safely.


Piramal Pharma Solutions' Ennore, India facility is a rare example of a site with long-standing expertise in commercial-scale ozonolysis. With more than a decade of experience, the Ennore team supports ozonolysis from laboratory development through commercial production.
The facility is equipped with both laboratory and large-scale ozonators, enabling seamless transition from R&D to manufacturing. Advanced process monitoring, experience with peroxide chemistry, and established safety protocols enable Piramal to reliably execute ozonolysis reactions at scale, including commercial campaigns up to 30 kg.
Case studies from Ennore demonstrate successful use of ozonolysis for challenging aldehyde formations and multi-step synthetic sequences, highlighting how controlled ozonolysis can unlock transformations that are difficult to achieve using other oxidation methods.
Ozonolysis remains one of the most efficient and precise oxidation reactions available to chemists, but its adoption at scale depends on rigorous process control and specialized infrastructure. Facilities with proven safety systems and hands-on experience are essential to unlocking its full potential. With established protocols and scalable capabilities, Piramal's Ennore site demonstrates how ozonolysis can be safely integrated into modern API manufacturing to support complex and high-value syntheses.
Ozonolysis offers high atom efficiency, precise bond cleavage, and minimal by-product formation without the use of heavy metals.
Ozonolysis is difficult to scale because the formation of unstable ozonides, peroxide intermediates, and the use of reactive ozone gas require specialized safety controls and equipment.
Yes, with proper process safety measures, monitoring systems, and experienced facilities, ozonolysis has been successfully implemented at multi-kilogram commercial scale.
